What Are the Characteristics of Hexagonal Boron Nitride?
Hexagonal boron nitride is also called white graphite. Similar to the hexagonal carbon mesh in graphite, nitrogen and boron in hexagonal boron nitride also form hexagonal mesh layers, which overlap each other to form crystals. Its crystal is similar to graphite, with diamagnetism and high anisotropy, and the crystal parameters are also quite similar. Due to its excellent properties, boron nitride is mainly used for refractory materials, semiconductor solid-phase doping sources, structural materials for atomic stacks, packaging materials for preventing neutron radiation, rocket engine constituent materials, high-temperature lubricants, and mold release agents. In this article, let's take a look at the characteristics of Hexagonal Boron Nitride.
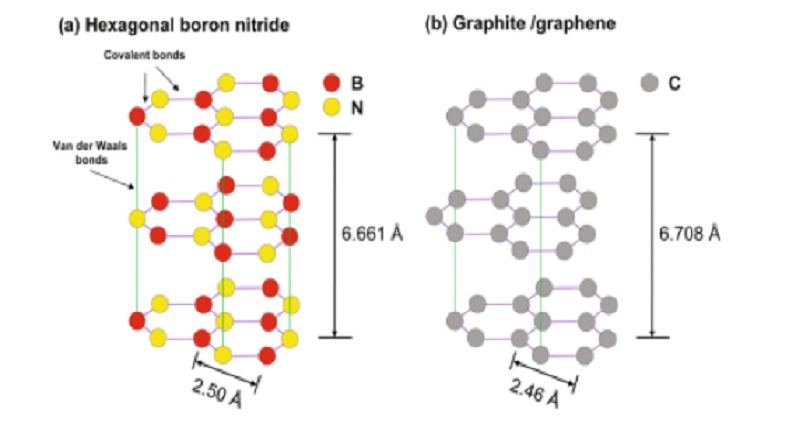
Characteristics of Hexagonal Boron Nitride
The characteristics of hexagonal boron nitride as a chemical additive are:
Hexagonal boron nitride (white graphite) is a loose, lubricating, moisture-absorbing white powder with a true density of 2.27/cm3, a Mohs hardness of 2, and low mechanical strength, but higher than graphite. It has no obvious melting point and sublimates at 3000℃ in 0.1Mpa nitrogen. Its stability in an oxygen atmosphere is poor, and its use temperature is below 1000°C.
Hexagonal boron nitride has a low expansion coefficient and high thermal conductivity, so it has excellent thermal shock resistance, and it will not be damaged even after hundreds of cycles at 1200-20°C. The expansion coefficient of BN is equivalent to that of quartz, but the thermal conductivity is 10 times that of quartz.
Hexagonal boron nitride is characterized by a good heat conductor and a typical electrical insulator. Its room temperature resistivity can reach 1016~1018. Even at 1000℃, the resistivity is still 104~106Ω.cm. The dielectric constant of BN is 3~5, the dielectric loss is (2~8) *10-4, and the breakdown strength is twice that of Al2O3, reaching 30-40 Kv/mm. Hexagonal boron nitride has good lubricity, oxidation resistance, corrosion resistance, insulation, thermal conductivity, and chemical stability. It can be used to manufacture TiB2/BN composite ceramics, high-grade refractory materials and super hard materials, horizontal continuous rolling steel separation rings, high-temperature resistant lubricants, and high-temperature coatings, and also a raw material for the synthesis of cubic boron nitride.
It is characterized by excellent chemical stability, neither wetting nor acting on most metal melts, such as steel, stainless steel, Al, Fe, Ge, Bi, Si, Cu, Sb, Sn, In, Cd, Ni, Zn, etc. Therefore, it can be used as a high-temperature galvanic couple protection cover, melting metal crucible, utensils, pipes for conveying liquid metal, pump parts, cast steel abrasive tools, and high-temperature electrical insulation materials. Due to the heat and corrosion resistance of BN, it can be used to manufacture high-temperature components, rocket combustion chamber linings, heat shields for spacecraft, and corrosion-resistant parts for magnetic flow generators.
Conclusion
Thank you for reading our article and we hope it can help you to have a better understanding of the characteristics of Hexagonal Boron Nitride. If you want to know more about Boron Nitride, we would like to advise you to visit Stanford Advanced Materials (SAM) for more information.
As a worldwide supplier of Hexagonal Boron Nitride products, Stanford Advanced Materials (SAM) has over two decades of experience in the manufacture and sale of Hexagonal Boron Nitride, offering customers high-quality Hexagonal Boron Nitride to meet their R&D and production needs. As such, we are confident that SAM will be your favorite Boron Nitride supplier and business partner.




